V-PLEX Quality

V-PLEX Immunoassays - Reproducible performance, consistent results.
Consistency and reproducibility are crucial for regulated environments and long term studies. MSD V-PLEX assays are designed to maximize reliability in results and confidence in the data. V-PLEX multiplex and single analyte assays are analytically validated to provide reproducible results from one kit lot to another, both within and between runs. The assays are developed in accordance with MSD’s stringent Quality Management System, which ensures analytical testing of assay sensitivity, precision, specificity, recovery, linearity, and repeatability in multiple sample types. Whether during development or manufacturing, V-PLEX assays are built using well-controlled processes that yield an end result with the highest standards of consistency.
-
Assay Development and Validation
-
Manufacturing
-
Quality Control Release
One of the primary goals during the development of V-PLEX assays is to determine and mitigate elements that can contribute to product variability.
Assay Development
Selection of the highest quality critical reagents is crucial for developing the most robust immunoassays. V-PLEX assay development starts with the selection of these critical reagents. Multiple candidate antibodies for each marker are screened using recombinant proteins, stimulated cells, and serum samples to confirm recognition of native protein.
Optimal antibody pairs are purified and analytically characterized to screen for aggregation and degradation, and to assess antibody purity. This extensive characterization is essential for confirming consistency between new lots of raw materials.
Multiplex panels are then designed according to biological relevance and endogenous analyte levels in samples. Optimization of the assays involves testing of the individual kit components as well as the complete kit as a unit. Antibody concentration, diluent formulation, buffer formulation, protocol, reagent and sample preparation, and manufacturing scale up are some of the parameters that are optimized. The final assembled kit is then validated and release specifications are set.
Assay Validation
V-PLEX assay development follows a rigorous, well-defined design control process. The assays are analytically validated according to the principles outlined in “Fit-for-Purpose Method Development and Validation for Successful Biomarker Measurement” by J. W. Lee, et al. (Pharm Res. 2006;23:312-28). Product validation of every V-PLEX panel includes manufacture of a minimum of three kit lots, using independent raw material lots when available. These are tested in multiple runs, across multiple days, and by multiple analysts. Parameters such as dynamic range, sensitivity, precision, specificity, recovery, linearity, and accuracy are optimized, and data from these lots are used to define specifications.
The data presented were generated in human V-PLEX assays.
Calibration Curve Optimization and Anchoring
Our goal in assay development is to generate assays that are capable of accurately measuring analyte concentrations across a broad linear dynamic range in order to accurately quantitate proteins at normal and disease state levels with minimal dilution.
For V-PLEX assays, calibration data is collected from >30 runs for each multiplex V-PLEX panel over several months.
All V-PLEX assay calibrators are anchored to a MSD reference calibrator to ensure consistency between lots. MSD calibrators are also evaluated against international reference standards (NIBSC/WHO), when available.
Limits of Quantification
During V-PLEX assay development, we determine and validate the analytical sensitivity (lower limit of detection or LLOD) for each assay. The LLOD is a calculated concentration corresponding to the signal 2.5 standard deviations above the background (zero calibrator). To ensure accurate quantification from lot to lot, we also establish the release specifications (CV ≤ 20%) for the functional limits of quantification (LOQ), which are verified for every manufactured lot. The quantitative range of the assay lies between the upper and lower LOQs.
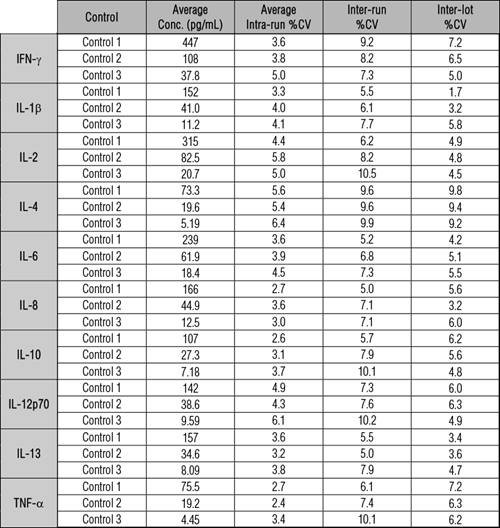
Reproducibility and Precision
Validation controls are measured across multiple plate lots for every V-PLEX assay to evaluate assay accuracy and precision. The results of control measurements typically fall within 20% of the expected concentration for each run.
Precision is determined as the % coefficient of variation (%CV) of the control replicates within a run, between runs, and across lots. We observe typical CVs for controls to be less than 10%, for both intra-run and inter-run.
Analyte levels were measured by 5 operators using a minimum of 3 replicates on 49 runs over 5 months.
Specificity
One of the key parameters in the development of a multiplex panel is ensuring high specificity of the capture and detection antibodies for each assay within the multiplex. Specificity of each V-PLEX assay is assessed during development as well as for each manufactured kit lot. Non-specific binding is typically less than 1% for all assays within a multiplex. Interference from other related biomarkers and proteins is also evaluated.
Evaluation of Matrix Effects
V-PLEX assays are validated for recovery and linearity in multiple, clinically relevant sample types such as serum, plasma, spinal fluid, and urine. Variability arising from matrix effects is evaluated by testing independent sets of samples on multiple plate lots, with a target recovery for each assay between 80% and 120%. These are then translated into release specifications.
Stability
V-PLEX assay kits have a long shelf life of 30 months from the date of manufacture. To ensure longevity of V-PLEX assays over the life of the product, the stability of all components and the fully assembled V-PLEX kits are evaluated at the assay development stage.
The stability of the lyophilized calibrator after reconstitution is assessed in real time over a 30-day period. Freeze–thaw testing of all frozen assay components is conducted through real-time and accelerated stability studies. V-PLEX assay validation includes a real-time stability study of complete kits for up to 54 months from date of manufacture.
MSD has over 20 years of experience in manufacturing high quality immunoassay products. MSD’s state-of-the-art 104,000 square foot manufacturing facility provides the ability to expand our production to meet the growing needs of our customers. Well-trained technical staff and the use of advanced equipment, including precise, low volume dispensers, automation systems, HPLC, DLS, and CIEF, enable the production of highly reliable and consistent immunoassay kits. Products are manufactured utilizing documented procedures controlled under MSD’s Quality Management System (QMS). MSD’s QMS has received ISO 9001:2008 certification for the design, development, manufacture, distribution, and service of instruments, plates, reagents, assays, and kits for research purposes.
V-PLEX kit manufacturing follows well-defined processes with functional testing incorporated for every component included in the kits. Prior to assembly, each V-PLEX kit component undergoes testing to ensure that the final product meets the specifications for reproducibility and consistency. Every step of the production process is carefully monitored for accuracy.
Plate Testing
Plate uniformity testing is performed with Protein A/G and calibrators to determine inter-and intra-plate consistency for every plate lot.

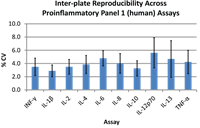
Data shown are for 35 lots of V-PLEX Proinflammatory Panel 1 (human) plates (Cat. No. K15049G). Intra-plate % CV and inter-plate % CV for all assays were observed to be less than 6% and less than 8%, respectively.
Component Testing
Antibodies, calibrators, and other reagents are tested against a reference lot to assess for adherence to specifications.
V-PLEX assay kits, once fully assembled, are functionally evaluated for dynamic range, limits of quantification, precision, and non-specific binding to ensure consistency and reproducibility. The lot-specific certificate of analysis (COA) shipped with each order attests to the quality of the product.
Kit QC
Certificate of Analysis
After component testing, kits are assembled and tested for:
- Performance against release specifications
- Precision and accuracy of control measurements
- Precision and accuracy of limits of quantification
- Calibration curve signal values
- Non-specific binding
- Each V-PLEX order is shipped with a lot-specific COA
- The COA outlines the kit release specifications for accuracy, precision, specificity, dynamic range, and sensitivity in addition to other kit-specific details such as component lot numbers and expiration dates
To date, over 400 lots of V-PLEX kits have been manufactured by MSD. The following plots illustrate the exceptional reproducibility of V-PLEX products. The % recovery and the % concentration CV of the controls tested on >81 lots of V-PLEX Proinflammatory Panel 1 (human) Kit manufactured over three years is shown.
Data shown represent 81 lots of V-PLEX Proinflammatory Panel 1 (human) kits (Cat. No. K15049G). Controls spanning the expected analyte range were measured. All controls were within the target range of +/-20% of the assigned concentration (left graph). The right graph shows the calculated concentration % CV, which is the variability of measured control concentrations across plates. The controls had low variability (CVs < 20%).