METHODS
Electrochemiluminescence Technology
MSD’s electrochemiluminescence detection technology uses SULFO-TAG labels that emit light upon electrochemical stimulation initiated at the electrode surfaces of MULTI-ARRAY and MULTI-SPOT microplates.

- Minimal background, combined with strong response to analyte, yields high signal-to-noise ratios.
- The stimulation mechanism (electricity) is decoupled from the response (light signal), minimizing matrix interference.
- Only labels bound near the electrode surface are excited, enabling non-washed assays.
- Labels are stable, non-radioactive, and directly conjugated to biological molecules.
- Emission at ~620 nm eliminates problems with color quenching.
- Multiple rounds of label excitation and emission enhance light levels and improve sensitivity.
Immuno-Oncology U-PLEX Protocol
The U-PLEX assay platform uses 10 unique linkers that specifically bind to individual spots, enabling simple and flexible creation of multiplex immunoassays.
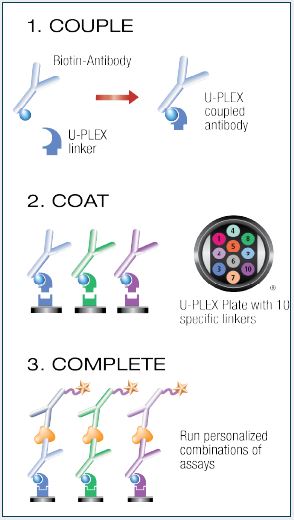
Couple and Coat the U-PLEX Plate:
- Add 200 µL of the biotinylated capture antibody to 300 µL of the assigned linker. Vortex. Incubate for 30 minutes.
- Add 200 µL of Stop Solution and vortex. Incubate for 30 minutes.
- Combine each U-PLEX-coupled antibody solution into a single tube and vortex. Add 50 µL of multiplex coating solution to each well.
- Incubate with shaking for 1 hour then wash the plate.
Complete the Assay:
- Add 50 µL of sample, calibrator, or control to each well.
- Incubate the plate for 2 hours, then wash the plate.
- Add 50 µL of detection antibody solution to each well.
- Incubate the plate for 1 hour, then wash the plate.
- Add 150 µL of MSD™ Read Buffer to each well and read the plate.
RESULTS
Assay Characteristics
Calibrator curves, lower limit of detection (LLOD), and upper limit of detection (ULOD), for 27 human immuno-oncology assays are shown below. LLODs were calculated from 3 runs each with >20 blank wells. Control samples for each assay showed expected precision and accuracy, with intra-run CVs less than 10%, inter-run CVs less than 25%, and recoveries largely within 70-130% of target concentrations (data not shown).

U-PLEX Biomarker Compatibility
Other assays in the U-PLEX product line were tested for compatibility with the new immuno-oncology assays using performance criteria such as dynamic range, sensitivity, sample detection, and non-specific binding between assays of <2%. 84 existing human U-PLEX assays were found to be compatible, creating a human U-PLEX immuno-oncology group with 111 biomarkers (see the table below) that can be used together in multiplexed panels.

Native Sample Testing
Immuno-oncology assays were evaluated for the ability to detect their respective analytes in human serum and EDTA plasma samples. Sample concentrations (pg/mL) were plotted with the top of curve (TOC) and LLOD for each analyte. Samples were diluted 4-fold except for BCMA/TNFRSF17, gp130 (soluble), HVACR2/TIM-3 and Tie-2 assays where samples were diluted 100-fold. FGF (basic) was not detected in human serum and plasma samples. All other analytes were detected irrespective of the type of matrix.

Human tissue lysate samples (5 µg) derived from different normal tissues (blue symbols) and tumor tissues (red symbols) were tested. Sample concentrations (pg/mL) were plotted with TOC and LLOD values for each analyte. All analytes were detected in most or all of the different tissue lysate samples.

Dilution Linearity
Serum and EDTA plasma samples were spiked with calibrator and diluted 2, 4, 8, and 16-fold before testing. Sample concentrations were normalized to the 4-fold sample dilution. For BCMA/TNFRSF17, gp130 (soluble), HAVCR2/TIM-3 and Tie-2, unspiked samples were diluted 50, 100, 200 and 400-fold. Sample concentrations were normalized to the 100-fold sample dilution. All analytes recovered within 70-130% in each type of sample.

Spike Recovery
Normal human serum and EDTA plasma samples were spiked with calibrator at 3 levels (high, mid, and low). Spike recovery values for the three spike levels were averaged and plotted. Recovery of most analytes was within 70-130% in each sample type.

Assay Specificity
Human immuno-oncology assays were evaluated for interference and competition with homologous and/or related analytes. In addition, the CTLA-4, PD1 and PD-L1 assays were tested for sensitivity to specific therapeutic antibody drugs. Assay interference was evaluated by comparing the recovery of a single mid-range analyte concentration in the presence of a wide range of concentrations of the potential interferent. Competition was evaluated by comparing human serum and EDTA plasma sample concentrations measured in singleplex and multiplex assay formats. Testing with the therapeutic antibody drugs Nivolumab and Pembrolizumab demonstrated that the PD1 (epitope 1) assay is more resistant to these drugs than the PD1 (epitope 2) assay. No unexpected assay interference or competition was observed.

CONCLUSIONS
Twenty-seven human assays were developed that provide sensitive and reliable measurements of traditional and emergent biomarkers associated with cancer and cancer immunotherapy. The assays can be used in singleplex and multiplex formats and include therapeutic antibody-sensitive and -insensitive PD-1 assays. Moreover, these assays can be run in combination with 84 additional biomarker assays, enabling researchers and drug developers to simultaneously measure immuno-oncology analytes along with cytokines, chemokines, and inflammatory markers.