2. Methods
MSD’s electrochemiluminescence detection technology uses SULFO-TAGTM labels that emit light upon electrochemical stimulation initiated at the electrode surfaces of MULTI-ARRAY and MULTI-SPOT microplates.
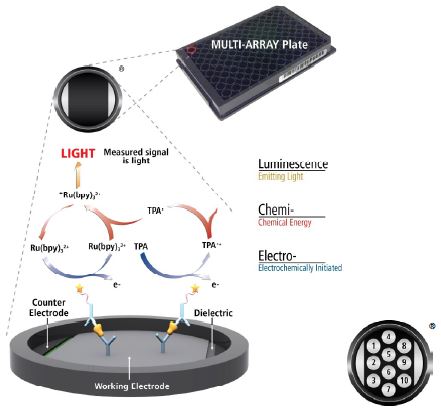
Electrochemiluminescence Technology
- Minimal non-specific background and strong responses to analyte yield high signal-to-background ratios.
- The stimulation mechanism (electricity) is decoupled from the response (light signal), minimizing matrix interference.
-
Only labels bound near the electrode surface are excited, enabling non-washed assays.
-
Labels are stable, non-radioactive, and directly conjugated to biological molecules.
- Emission at ~620 nm eliminates problems with color quenching.
-
Multiple rounds of label excitation and emission enhance light levels and improve sensitivity.
- Carbon electrode surface has 10X greater binding capacity than polystyrene wells.
- Surface coatings can be customized.
Quality Control Methods
Quality control testing of V-PLEX kits included measuring the precision (intra-plate and inter-plate %CVs) and accuracy (recovery of measured concentrations relative to expected values) for calibrators, LLOQ (lower limit of quantitation and ULOQ (upper limit of quantitation) samples, and control samples.
Limit of detection is a calculated concentration corresponding to the average signal at 2.5 standard deviations above the background (zero calibrator). LLOQ and ULOQ are established for the plate lot by measuring multiple levels of calibrator near the expected LLOQ and ULOQ. LLOQ and ULOQ are the lowest and highest (respectively) concentrations of calibrator tested which have %CVs of 20% or less, with recovered concentrations within 70-130%.
High, Mid, and Low QC controls are prepared by spiking recombinant calibrators into a non-human serum matrix.
The protocol utilized during the release of MSD human V-PLEX multiplex kits is shown at right.
Protocol
1. Wash plate 3 times with 150 µL/well Wash Buffer. Add 50 µL/well of sample (calibrator, controls, LOQ samples). Incubate at room temperature with shaking for 2 hours.
2. Wash plate 3 times with 150 µL/well Wash Buffer. Add 25 µL/well of 1X detection antibody solution. Incubate at room temperature with shaking for 2 hours.
3. Wash plate 3 times with 150 µL/well Wash Buffer. Add 150 µL/well of 2X Read Buffer T. Read on the MSD instrument.
3. Proinflammatory Panel 1 (human)
The following figures illustrate the quality control test results for 78 independently tested Proinflammatory Panel 1 (human) Kit lots. Testing was performed between May 2013 and September 2016 using 6 different control lots. Each data point represents the results compiled for a minimum of 5 plates.


4. Chemokine Panel 1 (human)
The following figures illustrate the quality control test results for 23 independently tested Chemokine Panel 1 (human) Kit lots. Testing was performed between July 2013 and January 2017 using 3 different control lots. Each data point represents the results compiled for a minimum of 5 plates.


5. Cytokine Panel 1 (human)
The following figures illustrate the quality control test results for 25 independently tested Cytokine Panel 1 (human) Kit lots. Testing was performed between May 2013 and March 2017 using 5 different control lots. Each data point represents the results compiled for a minimum of 5 plates.


6. Conclusion
A retrospective analysis of MSD’s rigorous quality control testing on more than 120 lots of human V-PLEX multiplex kits demonstrated high levels of precision and accuracy. Inter-plate concentration CVs were typically below 10%, with average measurements for intra-plate concentration CVs below 20% and measured recoveries within 20% of the expected concentration. The analysis confirmed the consistency and reproducibility of V-PLEX assay performance, demonstrating the suitability of these products for longitudinal studies.